 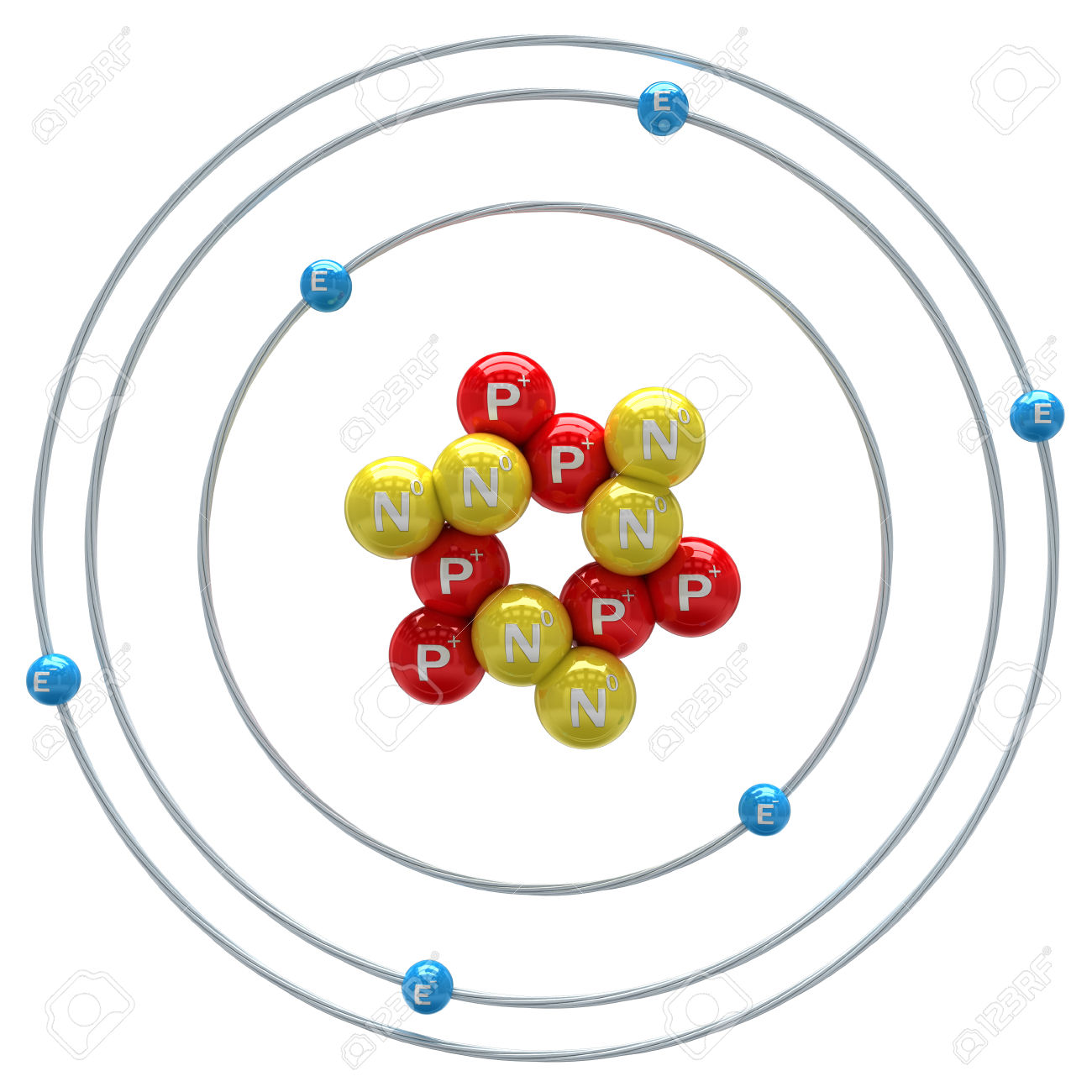
Molecules of fullerene show promise in a range of applications, including high-tensile-strength materials, unique electronic and energy-storage devices, and safe encapsulation of flammable gases, such as hydrogen. Because it conducts electricity but does not melt, graphite is also used for electrodes in electric furnaces and dry cells as well as for making crucibles in which metals are melted. In microcrystalline and nearly amorphous form, it is used as a black pigment, as an adsorbent, as a fuel, as a filler for rubber, and, mixed with clay, as the “lead” of pencils. Carbon as diamond is the most expensive and brilliant of all the natural gemstones and the hardest of the naturally occurring abrasives. Graphite, on the other hand, is a soft slippery solid that is a good conductor of both heat and electricity. Pure diamond is the hardest naturally occurring substance known and is a poor conductor of electricity. Get a Britannica Premium subscription and gain access to exclusive content. Before the discovery in 1779 that graphite when burned in air forms carbon dioxide, graphite was confused with both the metal lead and a superficially similar substance, the mineral molybdenite. The word carbon probably derives from the Latin carbo, meaning variously “coal,” “charcoal,” “ember.” The term diamond, a corruption of the Greek word adamas, “the invincible,” aptly describes the permanence of this crystallized form of carbon, just as graphite, the name for the other crystal form of carbon, derived from the Greek verb graphein, “to write,” reflects its property of leaving a dark mark when rubbed on a surface. Q-carbon is also synthetic, but scientists have speculated that it could form within the hot environments of some planetary cores. It was later found to occur naturally in tiny amounts on Earth and in meteorites. Fullerene was serendipitously discovered in 1985 as a synthetic product in the course of laboratory experiments to simulate the chemistry in the atmosphere of giant stars. Diamond and graphite occur naturally on Earth, and they also can be produced synthetically they are chemically inert but do combine with oxygen at high temperatures, just as amorphous carbon does. Other forms-such as carbon black, charcoal, lampblack, coal, and coke-are sometimes called amorphous, but X-ray examination has revealed that these substances do possess a low degree of crystallinity. Yet another form, called amorphous carbon, has no crystalline structure. A fourth form, called Q-carbon, is crystalline and magnetic. Spheroidal, closed-cage fullerenes are called buckerminsterfullerenes, or “buckyballs,” and cylindrical fullerenes are called nanotubes. A third form, called fullerene, consists of a variety of molecules composed entirely of carbon. Two of its well-defined forms, diamond and graphite, are crystalline in structure, but they differ in physical properties because the arrangements of the atoms in their structures are dissimilar. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!Įlemental carbon exists in several forms, each of which has its own physical characteristics.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today. 
Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.Demystified Videos In Demystified, Britannica has all the answers to your burning questions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
